|
The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. There are various synthetic isotopes of cobalt, but the most abundant naturally occurring stable isotope is the 59 Co. The melting point of cobalt is 1495 ☌ and its boiling point is 2927 ☌. Cobalt is a transition metal having bluish lustrous grey metallic surface. We know that as we scan down a group, the principal quantum number, n, increases by one for each element.Electron configuration chart of all Elements is mentioned in the table below. Physical properties of cobalt are mentioned below. General trends noted are increasing circle size moving from top to bottom in a group, with a general tendency toward increasing atomic radii toward the lower left corner of the periodic table. Sulfur has an atomic mass of 32.06 amu and has a density of 1.96 g/cm 3. The melting point of sulfur is 115.2 ☌ and its boiling point is 444.6 ☌. No spheres are provided for the noble or inert gas, group 18 elements. Hydrogen sulphide, which smells like rotten eggs is flammable and poisonous gas. You can also download the HD image of the periodic table with valence electrons. Beneath the molecule is the label, “I radius equals 266 p m divided by 2 equals 133 p m.” In figure b, a periodic table layout is used to compare relative sizes of atoms using green spheres. This is a periodic table with valence electrons labeled on it. The distance between the radii is 266 p m. Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm.” The fourth diatomic molecule is in purple. The distance between the radii is 228 p m. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. The distance between the radii is 198 p m. The second shell has 2 subshells: 1 s-orbital and 3 p-orbitals. This means that the first shell can hold 2 electrons. The first shell (of all atoms) has 1 subshell of s-orbitals containing 1 s orbital. The second diatomic molecule is in a darker shade of green. Every subshell has a of orbits s/p/d/f that can each hold 2 electrons each (one has the opposite spin of the other). Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. Usually it’s oxidation state is +1, but it can lose the electron and have a valence of -1. A lithium atom has one outer shell electron. Hydrogen is the lightest gas and due to this, it escapes the earth’s atmosphere easily. This electron arrangement indicates that the outermost orbit of chlorine element (Cl) has 7 electrons. Hence the chlorine element has electrons arrangement 2, 8, 7. Key groups include alkali metals, alkaline earth metals, halogens. The table is divided into metals, nonmetals, and metalloids, each with distinct properties. Elements in the same group share similar characteristics, like reactivity. Hydrogen is the most abundant element found in the universe and it is roughly 74. Now the atomic number of chlorine (Cl) is 17. The periodic table organizes elements into groups and periods based on their chemical and physical properties. It is believed that the hydrogen element was produced during the big bang. 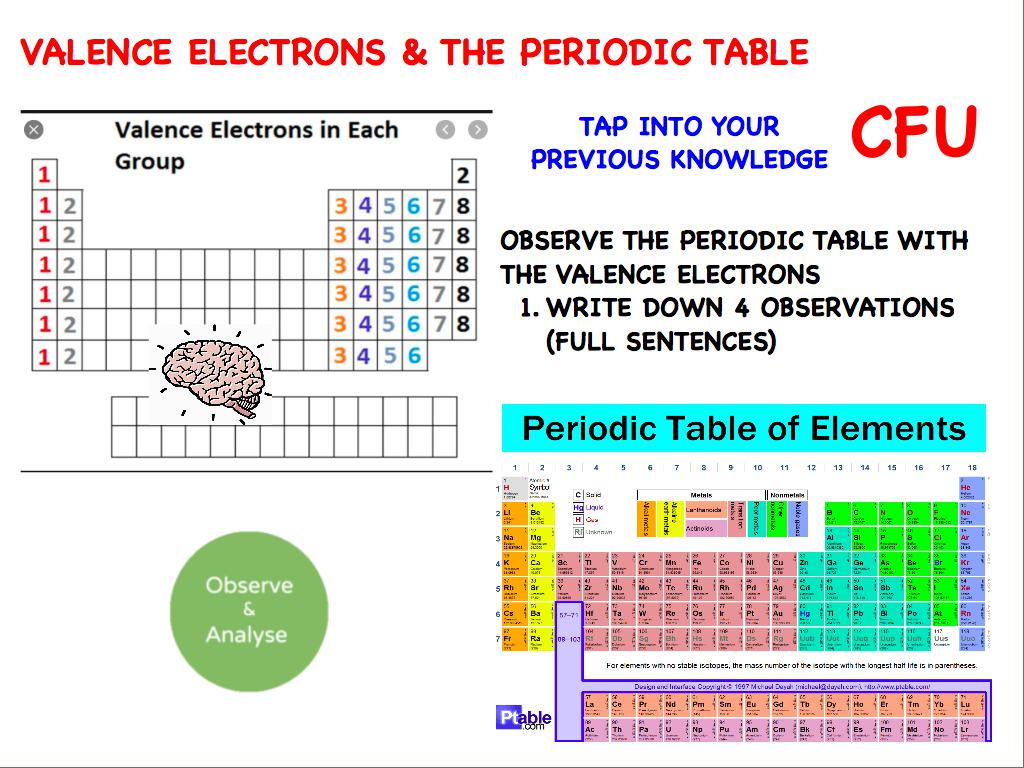
The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. The oxidation state tells how many valence electrons an atom accepts (negative number) or donates (positive number) to form a chemical bond. The chemical formula of water is H 2 O, which includes hydrogen atoms. Two spheres are pushed very tightly together. The first model, in light green, is used to find the F atom radius. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The general trend is that radii increase down a group and decrease across a period. :max_bytes(150000):strip_icc()/PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
(b) Covalent radii of the elements are shown to scale. 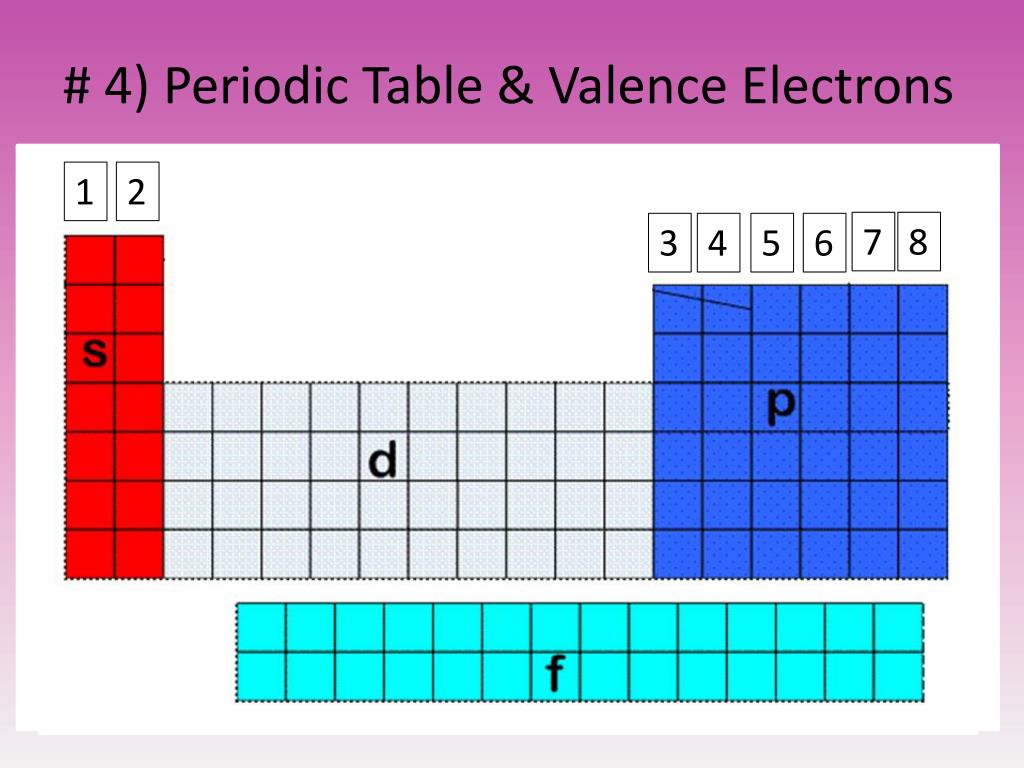
The atomic radius for the halogens increases down the group as n increases. \): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
